Clinical Hours: Mon-Fri, 8AM - 3PM(905) 493-5700
1885 Glenanna Road, Suite 220, Pickering, ON, L1V 6R6Exploring the surprising connection between porphyria, the disease behind vampire legends, and photodynamic therapy, a modern treatment for precancerous and cancerous skin lesions.

Few medical topics bridge folklore and clinical science as elegantly as the connection between porphyria, the metabolic disorder long associated with vampire mythology, and photodynamic therapy (PDT), a treatment now used to destroy precancerous and cancerous cells in the skin. The link between the two lies in a single family of molecules: porphyrins. These light-sensitive compounds, when accumulated abnormally, once condemned individuals to a life of sun avoidance and social isolation. Today, that same photosensitivity is deliberately harnessed to treat actinic keratosis, certain non-melanoma skin cancers, and other cutaneous pathologies.
The image of the vampire, a pale figure who shuns daylight, recoils from garlic, and exhibits an unusual relationship with blood, has persisted across cultures for centuries. In 1985, biochemist David Dolphin presented a hypothesis to the American Association for the Advancement of Science proposing that historical accounts of vampirism may have been influenced by individuals suffering from porphyria, a group of rare inherited or acquired disorders affecting heme biosynthesis.
Porphyrias arise from deficiencies in specific enzymes along the heme synthesis pathway. Heme is the iron-containing molecule that gives red blood cells their oxygen-carrying capacity. When one of the eight enzymes in this pathway is deficient or dysfunctional, intermediate products called porphyrins accumulate in the skin, blood, and organs. These accumulated porphyrins are intensely photosensitive.
The clinical features of certain porphyrias, particularly erythropoietic protoporphyria (EPP) and congenital erythropoietic porphyria (CEP), bear a striking resemblance to traits attributed to vampires in folklore:
Severe photosensitivity. Exposure to sunlight causes excruciating pain, blistering, and tissue destruction in affected individuals. Porphyrins deposited in the skin absorb light energy, primarily in the visible spectrum around 400 to 410 nm (the so called Soret band), and transfer that energy to molecular oxygen, generating reactive oxygen species (ROS) that damage cell membranes and surrounding tissue. Historically, affected individuals would have been compelled to avoid daylight entirely.
Facial disfigurement. In severe forms, chronic photodamage leads to scarring of the nose, ears, and lips, progressive destruction of facial cartilage, and receding gums that expose the teeth, creating a fang-like appearance. The combination of pallor, facial erosion, and nocturnal behaviour would have been deeply unsettling to pre-modern communities.
Red-stained teeth and urine. Porphyrin accumulation causes erythrodontia, a reddish-brown discolouration of the teeth that fluoresces under ultraviolet light. Urine may also turn red or dark brown. These visible markers would have further reinforced the perception of something unnatural or blood-related.
Garlic avoidance. Some researchers have suggested that allyl sulfide compounds in garlic may exacerbate porphyria symptoms by stimulating heme production and worsening the enzymatic bottleneck, though this aspect of the hypothesis remains debated.
While the porphyria-vampire theory has been criticised for oversimplification, and many historians argue that vampire folklore has complex social and cultural origins unrelated to any single medical condition, the biochemical parallels remain instructive. They illuminate how porphyrin photosensitivity produces dramatic clinical effects, and it is precisely this photosensitivity that modern medicine has turned into a therapeutic advantage.
The destructive power of porphyrins under light exposure, the very mechanism that causes tissue damage in porphyria, is the foundation of photodynamic therapy. The principle is straightforward: if porphyrins can be selectively concentrated in abnormal cells, and those cells are then exposed to a specific wavelength of light, the resulting photochemical reaction will destroy the targeted tissue while sparing surrounding healthy skin.
PDT requires three components, each of which is individually harmless: a photosensitising agent (the porphyrin precursor), a light source of a defined wavelength, and molecular oxygen present in the tissue.
The PDT process can be understood in three stages.
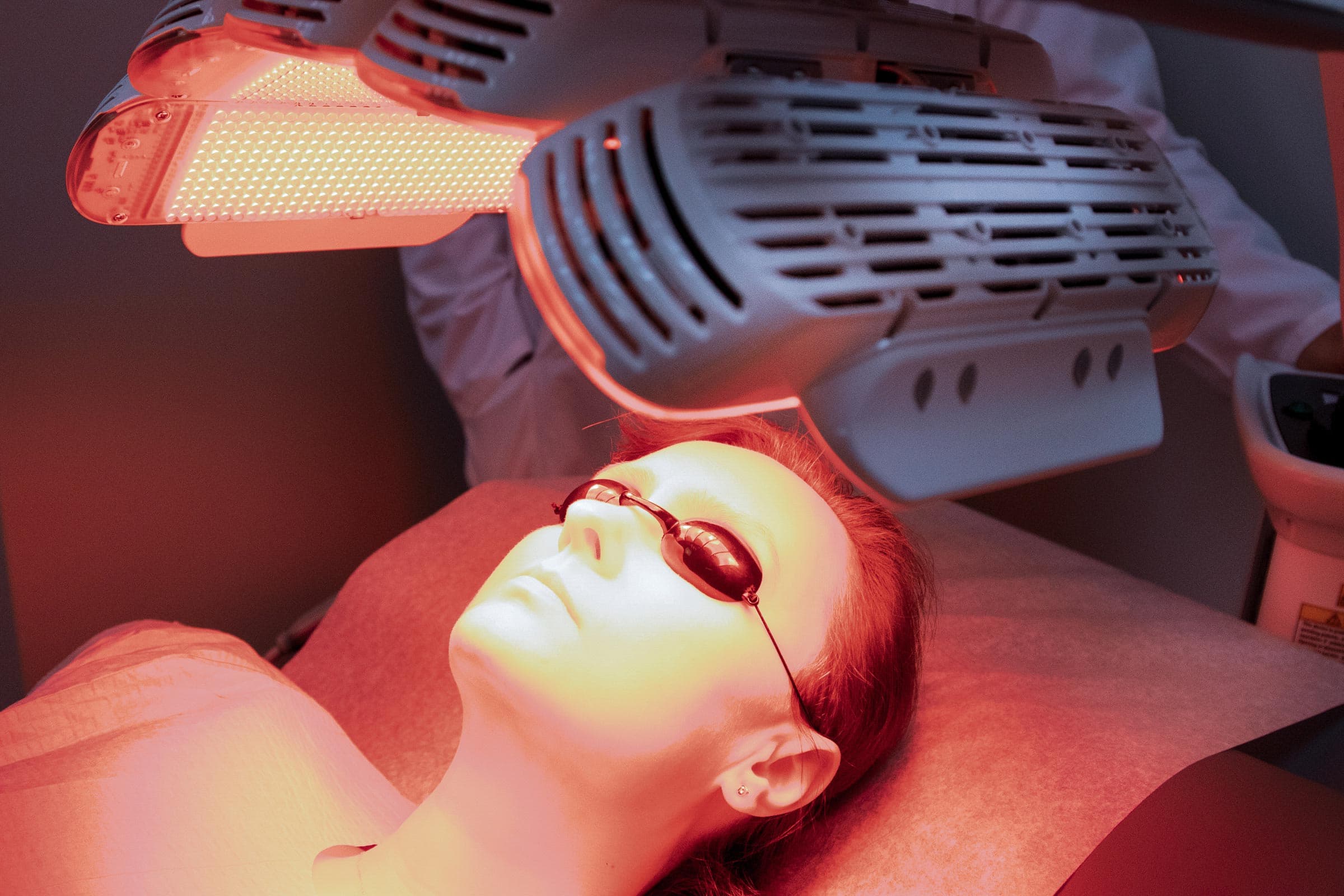
Stage 1: Application of the photosensitiser. A topical prodrug, most commonly 5-aminolevulinic acid (5-ALA) or its methyl ester (MAL), is applied to the treatment area. These compounds are not themselves photosensitive. They enter the heme biosynthesis pathway and are metabolised by cells into protoporphyrin IX (PpIX), a potent endogenous porphyrin. Critically, abnormal and rapidly proliferating cells, such as those found in actinic keratoses and superficial skin cancers, accumulate significantly higher concentrations of PpIX than normal cells. This selectivity occurs because dysplastic and neoplastic cells exhibit increased uptake of the prodrug, enhanced enzymatic conversion to PpIX, and reduced conversion of PpIX to heme due to decreased ferrochelatase activity.
Stage 2: Light activation. After an incubation period (that can be variable depending on the protocol), the treatment area is illuminated with a light source matching the absorption spectrum of PpIX. The most common light sources are the blue light around 410-415 nm, coinciding with the Soret band and red light at approximately 630-633 nm. When PpIX absorbs photons of the appropriate wavelength, it transitions from its ground state to an excited singlet state and subsequently to a longer-lived triplet state.
Stage 3: Cellular destruction. In the triplet state, the excited PpIX molecule transfers energy to ground-state molecular oxygen (O2), generating singlet oxygen (1O2) and other reactive oxygen species. These highly reactive molecules cause irreversible oxidative damage to cellular components, including lipid membranes, mitochondria, and the endoplasmic reticulum, triggering cell death through apoptosis and necrosis. Because PpIX is preferentially concentrated in abnormal cells, the destruction is targeted. Additionally, PDT damages the tumour microvasculature and triggers a local inflammatory immune response that contributes to the clearance of residual abnormal cells.
This is the same photochemical reaction that occurs in the skin of porphyria patients exposed to sunlight, but in PDT it is controlled, localized, and directed against diseased tissue.
Photodynamic therapy has established itself as a valuable treatment modality for several dermatological conditions, particularly those involving sun-damaged skin.
Actinic keratosis. Actinic keratoses are rough, scaly patches caused by cumulative ultraviolet radiation damage. They are classified as precancerous because a proportion can progress to squamous cell carcinoma if left untreated. PDT is particularly well suited for treating multiple or field-distributed actinic keratoses across broad areas such as the scalp, face, or forearms. Clinical clearance rates range from 70% to over 90% depending on the protocol and lesion thickness.
Superficial basal cell carcinoma. PDT is approved for the treatment of superficial basal cell carcinoma (sBCC), the most common form of skin cancer. For thin, superficial lesions, PDT offers an effective non-surgical alternative with excellent cosmetic outcomes, an important consideration when lesions are located on cosmetically sensitive areas such as the face. Two treatment sessions separated by one to two weeks are typically performed.
Bowen disease (squamous cell carcinoma in situ). This early, non-invasive form of cutaneous squamous cell carcinoma responds well to PDT, particularly in locations where surgical excision may result in significant scarring or functional impairment.
Field cancerisation. One of the unique advantages of PDT over lesion-directed therapies is its ability to treat field cancerisation, the concept that chronically sun-damaged skin contains widespread subclinical dysplasia beyond the visible lesions. By treating the entire affected field, PDT can address both clinically apparent actinic keratoses and subclinical abnormalities, potentially reducing the risk of future lesion development.
PDT offers several advantages over alternative treatments. It is non-invasive, produces superior cosmetic results compared to surgical approaches, can treat large surface areas in a single session, and is repeatable without cumulative toxicity. The selective uptake of the photosensitiser by abnormal cells provides an inherent margin of safety for surrounding healthy tissue.
The primary side effect is pain or a burning sensation during light exposure, which varies in intensity among patients and usually can be managed with cooling, local anaesthesia. However, the modern protocols such as “painless“ PDT avoid or drastically decrease these side effects. Treated areas typically develop erythema, oedema, and crusting that resolve over one to two weeks. Strict sun protection is required for 48 hours following treatment, as residual PpIX in the skin remains photoactive.
It is important to note that PDT is not appropriate for all skin cancers. Invasive squamous cell carcinoma, nodular basal cell carcinoma, and melanoma require different treatment approaches, typically involving surgical excision and, in some cases, adjuvant therapies. Accurate diagnosis through dermoscopic assessment and histopathological confirmation is essential before selecting PDT as a treatment modality.
While PDT represents an effective treatment option, prevention remains the most important strategy against skin cancer. The same ultraviolet radiation that drives actinic keratosis and non-melanoma skin cancer development is largely avoidable through consistent sunscreen use, protective clothing, and sun-avoidance behaviour during peak UV hours.
Regular skin cancer screening and self-examination enable early detection, when lesions are most amenable to non-invasive treatments such as PDT. Dermoscopic monitoring provides a systematic approach to tracking changes in sun-damaged skin over time, allowing clinicians to identify new or evolving lesions before they progress.
For patients who have already undergone treatment for actinic keratoses or non-melanoma skin cancer, ongoing post-treatment surveillance is recommended, as the underlying sun damage that produced the initial lesions places them at elevated risk for recurrence and the development of new lesions.
The story of porphyrins in medicine is one of remarkable transformation. The same photochemical property that may have condemned porphyria sufferers to lives of darkness and social ostracism, fuelling centuries of supernatural folklore, is now deliberately induced in diseased cells and activated with precision to treat and prevent skin cancer. What was once a source of suffering has become a therapeutic tool.
The vampire, it turns out, was never truly a creature of the night. It was a patient, misunderstood and feared, whose condition science had not yet explained. And the light that once caused that patient harm is now used, with intention and precision, to heal.
For patients with sun-damaged skin or concerns about precancerous lesions, a consultation can help determine whether photodynamic therapy or other treatment approaches may be appropriate.
Your feedback helps us improve our news and clinical insights
From medical dermatology to surgical procedures, our clinic provides comprehensive care for all skin, hair, and nail conditions.